Disease-first prioritisation
Programs are selected around unmet need, translational feasibility, and platform fit.
Reported incidence range across geographies
Based on current idursulfase dosage on a 50KG MPS II patient
Primarily affects males, carrier females may show symptoms
Multisystem accumulation leads to lifelong burden
Target identification, mechanism validation, lead optimization
IND‑enabling studies, formulation, toxicology
Phase I/II trials, biomarker validation, partnership expansion
Programs are selected around unmet need, translational feasibility, and platform fit.
Development is grounded in actionable disease biology rather than broad generic screening alone.
Knowledge gained from MPS II is intended to inform broader rare disease opportunities.
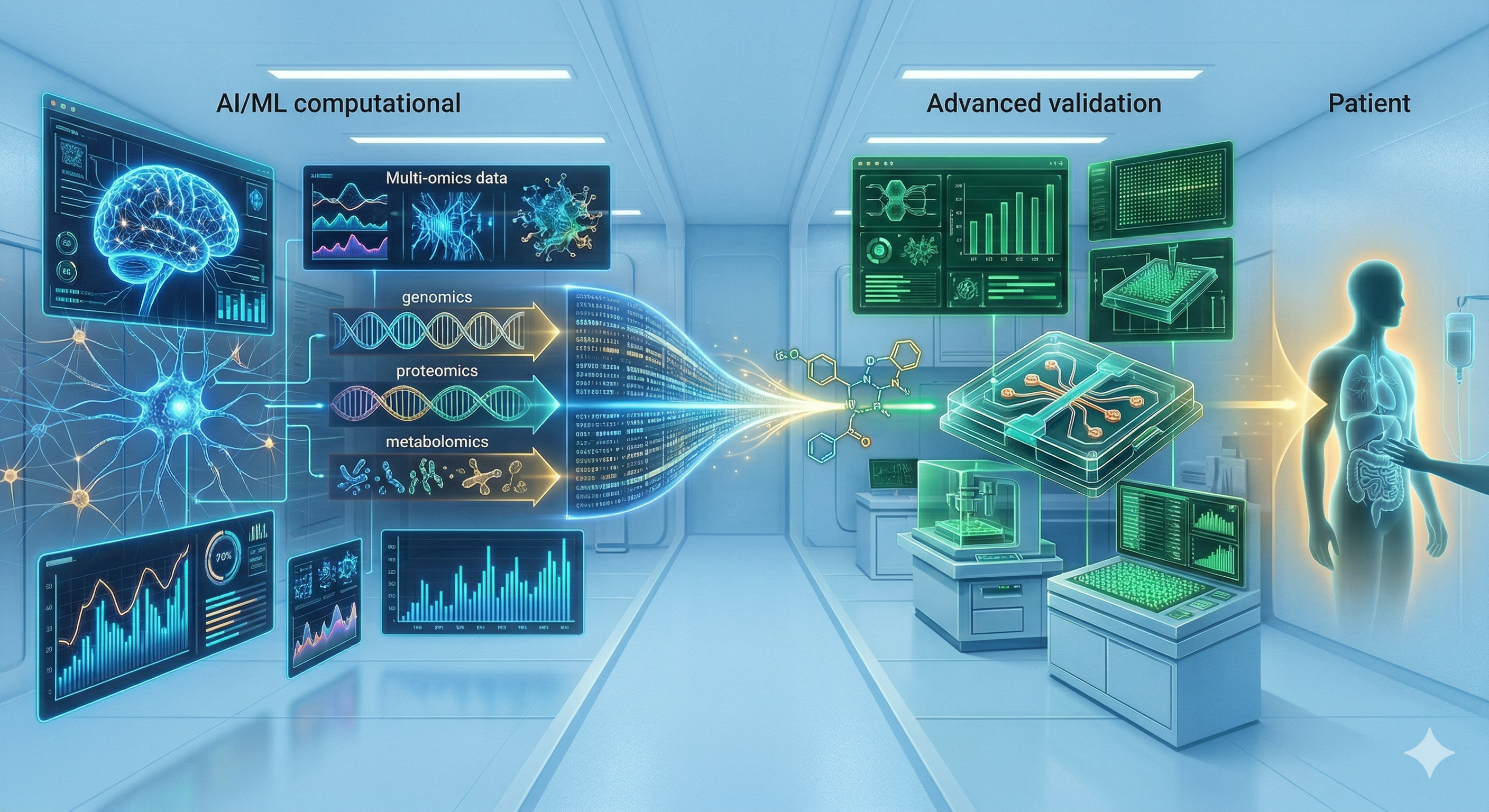
Bridging computational insight with advanced validation for better patient outcomes.